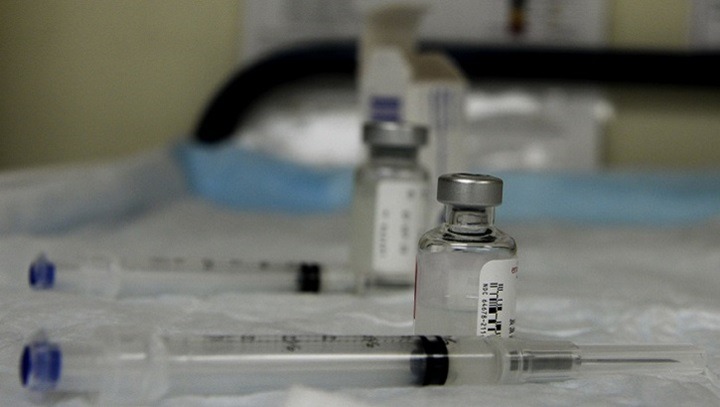
Standard protocols are very crucial for safeguarding human health. Before approving and allowing the use of a CORONAVIRUS (COVID-19) vaccine for trial in humans, regulators should evaluate safety and efficacy measures with a range of virus strains and in more than one animal model starting from lab mice to a monkey, etc. They should also have enough preclinical evidence that supports the experimental vaccines for preventing the infection, which means that we will probably have
A Phase 1 of clinical trial has begun at Kaiser Permanente Washington Health Research Institute in Seattle to evaluate an investigational vaccine designed to protect against the novel Coronavirus disease (COVID-19) The NIAID, a part of the National Institutes of Health, is funding the trial. The study is just to analyze the safety level by evaluating different doses of the experimental vaccine and its ability to induce an immune response in patients. It is the first
The outbreak of CORONAVIRUS and severe travel restrictions to stop its spread has taken a huge leap on the world’s second-largest economy and disrupted several industries dependant on Chinese supplies. India procures about 70 percent of API (active pharmaceutical ingredient) from China. The Indian pharma inventories have stock for a very short term and if travel restrictions continue conditions will worsen. Indian government is taking major steps to encourage domestic production of API’s to counter
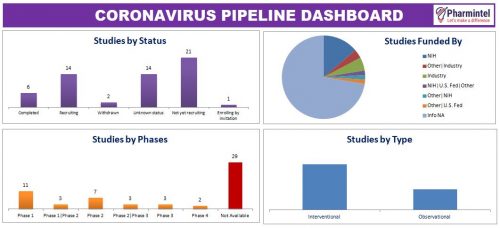
The novel coronavirus outbreak in China, Wuhan has been termed as Covid-19, is spreading rapidly to other countries and millions of lives are endangered. This pandemic has catalyzed the development of novel coronavirus vaccines in the industry and several companies are coming up with treatment and therapy for this deadly virus. Recently Favilavir, an anti-viral drug, as a treatment for Coronavirus has been approved by the National Medical Products Administration of China. The drug reportedly has
Sales Target
Category: Blog
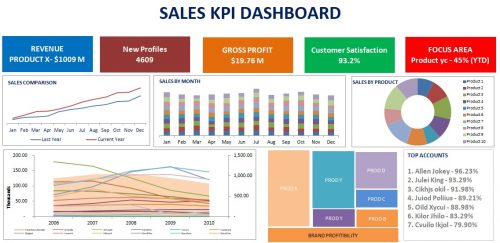
It’s an important factor for sales personals to accomplish sales target. Sales target plays an important role in companies’ annual turnover, market share, profitability, number of new account and renewal of existing contracts, etc. Sales target is the driving factor of company’s growth but under performing sales employees make it vulnerable. For most of the sales employees it becomes a tedious job to meet targets which are exceeding limits. To understand how to beat sales